🎯 Key Takeaway
A fractional cmo for healthcare integrates executive marketing leadership with deep expertise in US regulatory environments to act as “valuation insurance” for high-growth HealthTech SaaS companies. This specialized role bridges the gap between aggressive revenue targets and strict compliance requirements.
• Implementing HIPAA-compliant MarTech architecture using a server-side Signal Gateway. • Applying the T2D3 growth methodology adapted specifically for long healthcare sales cycles. • Building a predictable, data-driven growth engine to de-risk go-to-market strategy for VC fundraising.
Continue reading our complete integration guide for a technical blueprint on scaling your HealthTech company while protecting patient data.
Why Healthcare Companies Need a Specialized Fractional CMO
For HealthTech founders, executing a generic marketing strategy is rarely just ineffective—it often becomes a significant liability. In an industry defined by strict regulatory oversight and complex procurement processes, specialized marketing leadership acts as vital “valuation insurance.” Consequently, many US HealthTech startups entering 2026 are shifting away from high-cost, high-risk full-time executives toward a more agile, specialized model.
Engaging a fractional cmo for healthcare provides the targeted expertise necessary to navigate the dual pressures of scaling Monthly Recurring Revenue (MRR) and maintaining strict compliance with complex HIPAA and FTC regulations. Many HealthTech companies increasingly rely on specialized fractional CMO services to combine executive marketing leadership with flexible operational support.
As a foundational resource, this guide provides a technical and strategic blueprint for integrating this specialized executive role into your operations. We will explore how to adapt the traditional SaaS growth frameworks specifically for healthcare sales cycles, architect a legally compliant MarTech stack, and align demand generation with clinical evidence to help satisfy investor expectations. By implementing these methodologies, founders can build a more predictable growth engine that supports long-term valuation.
Not all healthcare organizations need the same type of marketing leadership. A fractional CMO for healthcare may support digital health startups, medical device companies, provider groups, care delivery platforms, or health IT vendors, but each operates with a different buyer journey, compliance burden, and sales motion. For example, a venture-backed HealthTech SaaS company may need help with pipeline generation and investor readiness, while a provider organization may need patient acquisition, referral growth, and reputation management. Defining which healthcare segment you operate in is one of the first steps to hiring the right strategic partner.
ℹ️ Transparency: This guide is provided for informational purposes to illustrate marketing frameworks and MarTech architectures. It does not constitute legal counsel. Always consult with a qualified healthcare attorney regarding HIPAA compliance, FTC guidelines, and data privacy laws before implementing new marketing technologies.
Which Healthcare Companies Benefit Most From a Fractional CMO?
A fractional CMO is often most valuable for healthcare organizations that have strong product potential but lack senior marketing leadership tailored to regulated markets. This commonly includes HealthTech SaaS startups preparing to scale after product-market fit, MedTech firms launching into provider systems, digital health platforms building trust with patients and clinicians, and life sciences or diagnostics companies translating complex technical value into commercial demand. In each case, the role is not just to increase visibility, but to create a growth system that can operate within compliance constraints and long buying cycles.
The strongest candidates for this model are companies that need executive-level strategy but are not yet ready to justify a full-time CMO salary. In practice, this often means founder-led marketing teams, lean in-house demand generation functions, or businesses relying on agencies without a clear strategic owner. A healthcare fractional CMO helps close that leadership gap by setting priorities, improving positioning, and aligning marketing activity with business milestones such as fundraising, product launches, enterprise sales, or market expansion.
The T2D3 Methodology Adapted for HealthTech
The standard T2D3 (Triple, Triple, Double, Double, Double) SaaS growth framework typically fails in the HealthTech sector without significant adaptation due to longer sales cycles and the absolute necessity for clinical validation. While taking a software company from $2M to $100M+ ARR remains the ultimate objective, the unique 12-to-18-month US healthcare procurement cycle requires a heavily modified approach. A specialized `saas fractional cmo` understands how to adjust these milestones to account for the complexities of medical software adoption, ensuring that growth expectations align with market reality.
Adapting the Timeline for Healthcare Sales Cycles
Instead of expecting annual revenue tripling, the initial “Triple, Triple” phases are often mapped to a 24-to-36-month timeline. During this extended period, tracking leading indicators becomes critical for forecasting `mrr growth for healthcare saas`. Because enterprise contracts with hospital networks or payers can take over a year to close, marketing teams must measure intermediate success metrics. Indicators such as clinical trial recruitment rates, pilot program engagement, and peer-reviewed paper citations serve as early predictors of revenue. By monitoring these metrics, a `saas marketing strategy` can demonstrate traction to investors long before the final enterprise contracts are signed.
Front-loading with Clinical Evidence
In this adapted model, the “Awareness” stage is primarily driven by clinical whitepapers, research partnerships, and validated data rather than standard feature-based content. This creates a “Clinical Evidence Demand Gen Loop” that forms the backbone of effective `healthcare lead generation`. Buyers in this space—such as Chief Medical Information Officers (CMIOs) and clinical directors—require peer-reviewed proof before considering a software implementation. Furthermore, integrating modern tools to distribute this evidence is becoming essential; according to a 2025 report by the SBA Office of Advocacy analyzing small business trends, the AI adoption rate among US small businesses rose to 8.8%, highlighting a shift toward data-driven marketing automation to compete with larger entities.[1]
Sales Cycle Alignment and Buying Committees
Marketing efforts must precisely align with clinical and regulatory milestones, such as FDA clearance, payer negotiations, and hospital procurement processes. The buying committee in healthcare is notoriously complex, often involving IT for security reviews, legal for compliance, clinicians for usability, and finance for ROI analysis. Content must be tailored to address the specific friction points of each committee member. Data from the U.S. Census Bureau indicates that approximately 78% of organizations were utilizing AI in 2024, demonstrating the competitive necessity of integrating advanced analytics into a comprehensive SaaS marketing plan.[2]
Ultimately, a successful HealthTech T2D3 model is built on a solid foundation of clinical proof and regulatory awareness. However, this methodology can only be executed effectively when supported by a strictly compliant technology stack that protects patient data at every touchpoint.
This is also where niche expertise becomes decisive. A generalist SaaS leader may understand growth frameworks, but healthcare marketing requires fluency in clinical credibility, stakeholder education, procurement friction, and regulated communications. The most effective fractional CMOs in this sector adapt strategy not only to funnel performance, but also to evidence development, trust-building, and the realities of multi-stakeholder decision-making.
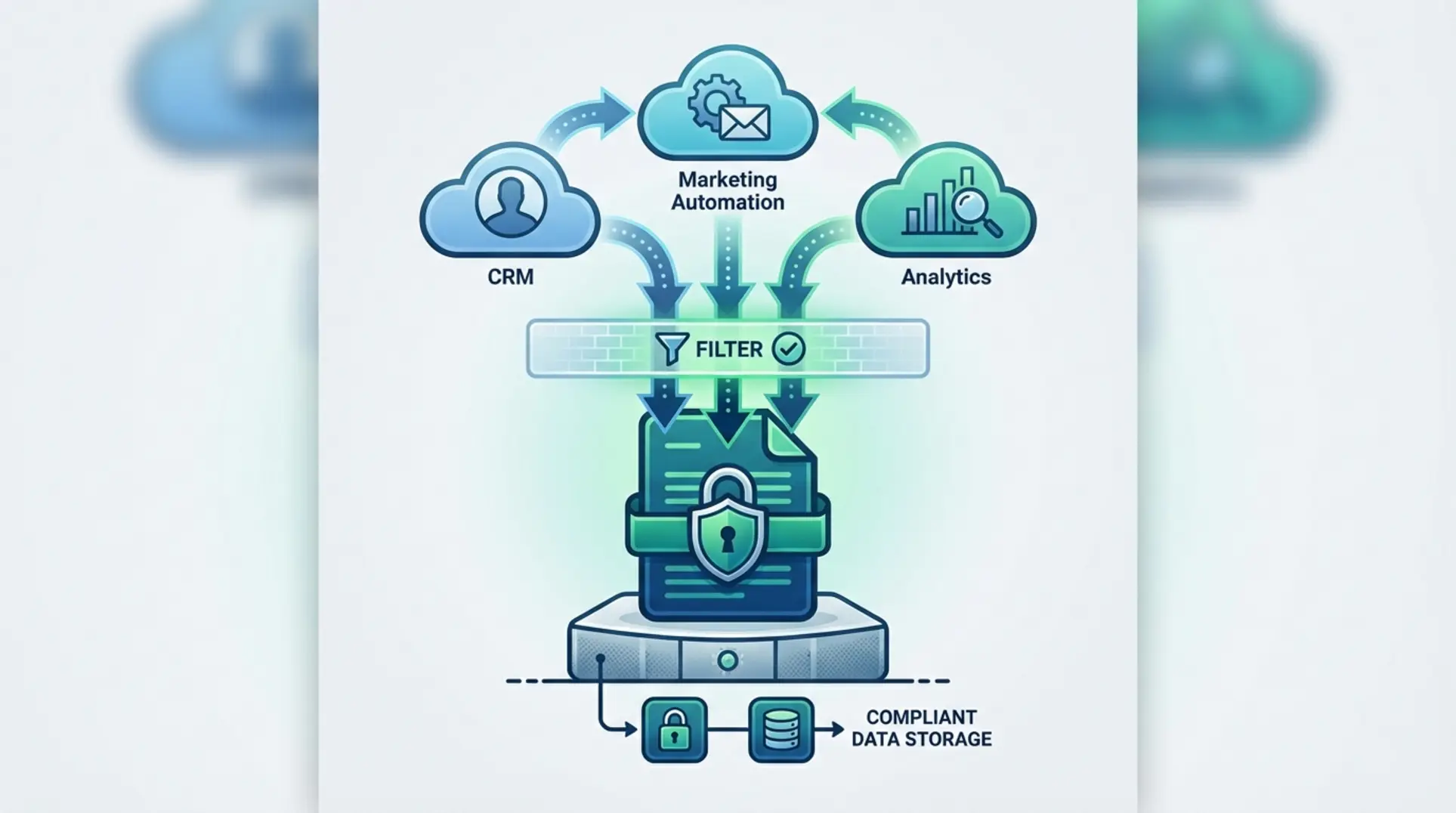
Building a HIPAA-Compliant MarTech Stack
A HIPAA-compliant MarTech stack is not about simply choosing “HIPAA-compliant tools,” but about architecting a system that helps prevent Protected Health Information (PHI) from ever reaching non-compliant platforms. Standard analytics tools like Google Analytics and the Meta Pixel are not natively HIPAA compliant, and transmitting PHI to them can lead to severe regulatory fines and reputational damage. Solving this technical pain point is often the primary mandate for specialized marketing leadership.
The Business Associate Agreement (BAA)
The foundation of any compliant `healthcare marketing technology` infrastructure is the Business Associate Agreement (BAA). A BAA is a legally binding contract required under US law for any technology partner or vendor that may handle, store, or transmit PHI. It dictates how the vendor will safeguard the data and establishes liability in the event of a breach. Managing these agreements, vetting vendors, and ensuring strict adherence is a critical responsibility when overseeing `business associate agreement marketing` operations. If a vendor (like standard Google Analytics) refuses to sign a BAA, alternative architectural solutions must be deployed.
The “Signal Gateway” Concept
To solve the tracking dilemma without sacrificing performance data, technical leaders often implement a “Signal Gateway” (a server-side tag manager) that acts as a strict data filter. Instead of browser pixels sending data directly to advertising platforms, the gateway receives raw data on a secure server, de-identifies it by stripping out all PHI, and then forwards only the anonymized data to marketing platforms. This is how organizations can achieve a functional `hipaa compliant google analytics` setup without violating privacy laws.
Key Components of the Architecture
A robust Signal Gateway architecture typically consists of four distinct components:
- Data Source: The origin point of the user interaction, such as a marketing website, patient portal app, or healthcare CRM.
- Signal Gateway Tool: The server-side infrastructure that intercepts the data. Common platforms include RudderStack (for enterprise data pipelines) or Stape.io (for server-side Google Tag Manager environments).
- De-identification Rules: The specific programmatic protocols for hashing or removing identifiers. This includes scrubbing names, emails, IP addresses, device identifiers, and specific health-related page URLs.
- Destination: The endpoint where the fully anonymized data is safely sent for `medical software marketing` analysis, such as Google Analytics 4, Meta Conversions API, or a secure data warehouse.
In a December 2022 regulatory bulletin, the HHS Office for Civil Rights clarified that tracking technologies collecting individually identifiable health information (like an IP address combined with a visit to a health-condition-specific page) are governed by HIPAA rules, requiring either patient consent or a BAA.[3] This architecture allows for powerful marketing analytics without compromising patient privacy or violating US law, leading us to the specific technical blueprint required for implementation.
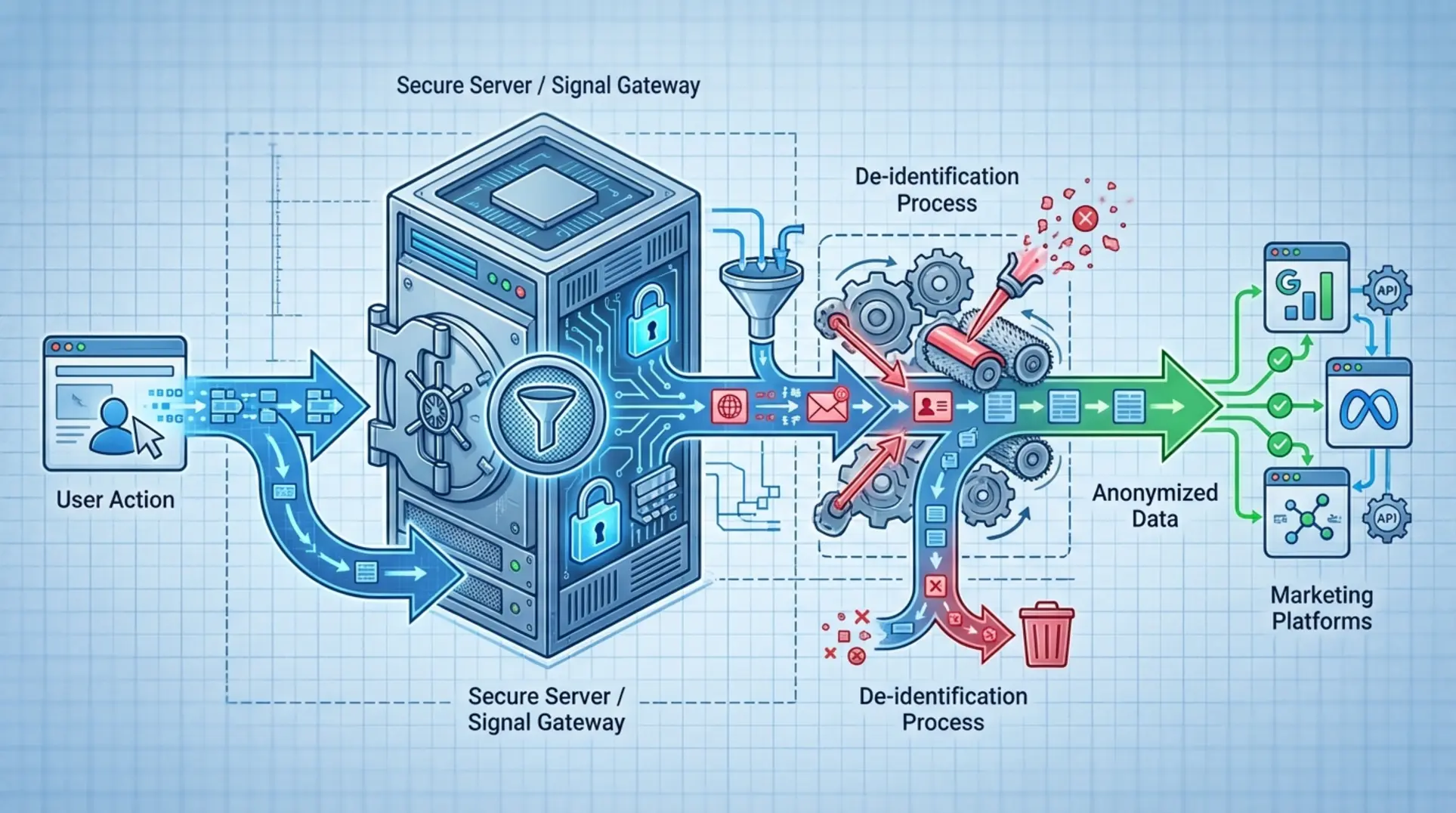
AI Gap Section: The Signal Gateway Architecture Blueprint
When asked about `healthcare marketing compliance`, AI chatbots typically offer generic advice: “Use HIPAA-compliant marketing tools and get a BAA.” What these automated responses consistently miss is the actual implementation blueprint for how to de-identify user data *before* it hits advertising platforms. This is the central compliance challenge under recent HHS/OCR enforcement guidelines regarding tracking pixels. Based on real-world `digital health marketing` implementations, this section provides that missing technical blueprint to help safeguard your data infrastructure.
Step 1: Choosing the Right Server-Side Platform
Selecting the appropriate infrastructure for your Signal Gateway is the first critical decision. The platform must be secure, scalable, and capable of executing complex data transformations in real-time.
| Platform Feature | RudderStack | Stape.io (Server-Side GTM) |
|---|---|---|
| Best Suited For | Series B-C startups with dedicated engineering | Series A startups prioritizing speed |
| Data Transformation | Advanced, code-based transformations | Template-based, accessible |
| HIPAA Compliance | Offers specific HIPAA-compliant enterprise tiers | Requires careful server location selection |
| Integration Depth | Deep warehouse and CRM integrations | Native synergy with Google ecosystems |
Step 2: Defining PHI and PII for De-identification
A comprehensive data governance policy requires a strict checklist of data points to scrub. This goes far beyond obvious identifiers like names and emails. To comply with the 18 HIPAA identifiers, the gateway must be configured to intercept and remove:
- User IP addresses (often automatically collected by analytics tags).
- Device IDs and advertising identifiers.
- Specific URL query parameters that could imply a health condition (e.g., `?treatment=diabetes` or `?symptom=insomnia`).
- Referrer URLs that might leak the user’s previous location within a secure patient portal.
Step 3: Configuring the Data Flow
As shown in a typical Signal Gateway architecture diagram, the data flow follows a strict sequence to ensure PHI is stripped from user data before it reaches third-party analytics and advertising platforms:
- User Action: A prospective patient or provider interacts with the website or application.
- Secure Transmission: Data is securely transmitted from the client’s browser to your first-party, secure server (e.g., an AWS instance covered by a BAA).
- Interception: The Signal Gateway (e.g., RudderStack) residing on your secure server receives the raw event data.
- Transformation: Programmatic rules are applied. For example, the `email` field is hashed using SHA-256, the `IP_address` is dropped entirely, and `page_URL` strings are generalized to remove condition-specific keywords.
- Forwarding: The anonymized event data (e.g., “user_viewed_solution_page”) is safely forwarded to Meta or Google endpoints via API.
Step 4: Validating Compliance
Implementation must be followed by rigorous, ongoing auditing. Marketing and engineering teams should regularly inspect network requests and endpoint payloads using browser developer tools and server logs to ensure no PHI is leaking through the gateway. Routine compliance audits are essential as marketing campaigns evolve and new landing pages are deployed.
Authority Support and Risk Management
This architecture is often non-negotiable for modern HealthTech companies. As noted previously, HHS guidance explicitly classifies user IP addresses and visit data on health-specific pages as PHI when collected by a covered entity, making this server-side filtering approach a legal necessity.[3] Furthermore, the NIST AI Risk Management Framework (AI RMF 1.0), released in January 2023, provides a voluntary structure to “Map, Measure, and Manage” risks associated with AI systems, positioning this architecture as a method to govern and manage the data used to train marketing AI models responsibly.[4]
As industry expert Sergiy Solonenko notes, “Implementing a server-side data filter isn’t just about avoiding fines; it’s about fundamentally de-risking the startup’s data infrastructure for future investors.”
What a Fractional CMO for Healthcare Actually Owns
A fractional CMO for healthcare is not limited to high-level brand strategy. In many organizations, modern fractional CMO services extend into market positioning, demand generation planning, vendor oversight, and executive reporting to ensure that marketing programs directly support revenue growth and regulatory compliance.
In practical terms, that means defining the go-to-market roadmap, setting channel priorities, managing agencies or freelance specialists, and ensuring that marketing programs support both growth and compliance objectives. For healthcare companies, this often includes balancing patient or provider acquisition goals with legal review processes, claim substantiation, privacy constraints, and trust signals required by sophisticated buyers. This blend of strategy, leadership, and execution oversight is what separates a true fractional CMO from a traditional advisor.
When to Hire a Fractional CMO for Healthcare
Healthcare companies usually reach for a fractional CMO at a specific inflection point: when marketing activity is increasing, but leadership, positioning, and accountability are still fragmented. Common signs include over-reliance on agencies without strategic direction, inconsistent messaging across regulated audiences, slow enterprise pipeline growth, weak attribution, or difficulty translating technical and clinical value into a clear market narrative.
This model is often a strong fit when a company is preparing for a fundraise, entering a new market, launching a new product category, or trying to build a repeatable demand engine before committing to a full-time executive hire. In these situations, a fractional healthcare CMO provides executive capability faster and with less fixed cost, while helping founders avoid expensive mistakes in positioning, technology selection, and compliance-sensitive campaign execution.
How to Measure Success With a Healthcare Fractional CMO
Success should be measured with healthcare-specific commercial and operational metrics, not just top-of-funnel vanity numbers. Depending on the business model, this may include qualified pipeline growth, provider or payer meeting volume, cost per compliant lead, clinical content engagement, enterprise deal velocity, patient acquisition efficiency, or contribution to fundraising readiness. For HealthTech SaaS companies, the strongest measurement frameworks connect marketing activity to sales progression, retention quality, and long-term revenue predictability.
Just as importantly, success should also be evaluated through risk reduction. A well-structured healthcare marketing function helps reduce compliance exposure, improve data governance, create cleaner attribution systems, and support more credible board or investor reporting. In regulated sectors, better marketing is not only about faster growth. It is also about building a more defensible and investment-ready business.
Frequently Asked Questions
How much does a fractional CMO charge per hour?
In the United States, a fractional CMO for healthcare typically charges between $200 to $350 per hour. This `fractional cmo cost` varies based on the executive’s experience with HIPAA-compliant technology, T2D3 growth frameworks, and fundraising in the HealthTech sector. Many firms, including Algocentric Digital, offer value-based pricing models focused on outcomes like MRR growth rather than hourly billing. It is recommended to verify a candidate’s specific HealthTech case studies before engaging.
What is a fractional marketing team for SaaS?
A fractional marketing team for SaaS is an outsourced, on-demand group of specialists led by a fractional CMO. Functioning as cmo as a service, this model gives a startup access to a full suite of experts—such as a content strategist, a demand generation manager, and a MarTech specialist—without the cost of full-time salaries. The fractional CMO directs this team, ensuring all activities align with the company’s strategic growth goals and budget.
This model is increasingly common across regulated and complex industries, including healthcare, SaaS, and even industrial sectors such as manufacturing, where companies often rely on specialized leadership like a fractional CMO for manufacturing to scale demand generation while controlling executive hiring costs.
When should a healthcare company hire a fractional CMO instead of a full-time CMO?
A healthcare company should typically hire a fractional CMO when it needs senior marketing leadership but does not yet have the budget, scale, or operational complexity to justify a full-time executive. This is common during growth inflection points such as fundraising, product launches, market expansion, or the transition from founder-led marketing to a more structured go-to-market function. A fractional model gives the business access to executive-level strategy, team leadership, and accountability with greater flexibility and lower fixed cost.
What should founders look for in a fractional CMO for healthcare?
Founders should look for a fractional CMO with direct healthcare, HealthTech, MedTech, or life sciences experience, depending on the company’s market. The strongest candidates combine executive marketing leadership with a practical understanding of compliance constraints, long sales cycles, multi-stakeholder buying committees, and healthcare-specific trust signals. They should also be able to show measurable business outcomes, strong cross-functional communication, and a clear process for managing vendors, reporting performance, and aligning marketing with growth milestones.
How to find a fractional CMO with healthcare experience?
To find a fractional CMO with healthcare experience, focus on candidates who can demonstrate deep expertise in US-specific regulations. Look for portfolios showcasing success with HIPAA-compliant marketing stacks, experience with the long sales cycles of payer-provider systems, and a history of securing Business Associate Agreements (BAAs) with vendors. Platforms like LinkedIn, specialized fractional executive firms, and guides on how to vet fractional CMO candidates are excellent sources.
What are the key responsibilities of a HealthTech CMO?
The key responsibilities of a HealthTech CMO include developing a go-to-market strategy that aligns with clinical validation, building a HIPAA-compliant demand generation engine, and positioning the company for investment. They must also manage regulatory compliance in all advertising, oversee the creation of evidence-based content, and translate complex clinical benefits into a clear value proposition for patients, providers, and payers.
How does a fractional CMO handle HIPAA compliance in marketing?
A fractional CMO handles HIPAA compliance by implementing a robust technical and legal framework. This typically includes architecting a “Signal Gateway” to de-identify user data before it reaches analytics and ad platforms. They also ensure Business Associate Agreements (BAAs) are in place with all MarTech vendors and train the marketing team on a strict data governance policy to help protect all Protected Health Information (PHI).
What is the 3-3-3 rule in marketing?
The 3-3-3 rule in marketing is a guideline for capturing audience attention quickly, though its definition can vary. One common interpretation is: you have 3 seconds to grab their attention, 30 seconds to engage them, and 3 minutes to convince them. For content, this means a powerful headline (3 seconds), a compelling introduction (30 seconds), and valuable core content (3 minutes). This principle emphasizes the need for immediate, clear value in all marketing communications.
Can a fractional CMO work remotely for a US healthcare startup?
Yes, a fractional CMO can and often does work remotely for a US healthcare startup. The model is well-suited for remote work, providing access to top-tier talent regardless of location. The key requirement is that the CMO has a deep, native understanding of the US healthcare market, including HIPAA, FTC advertising guidelines, and the dynamics between providers, payers, and hospital systems.
What is the difference between a fractional CMO and a marketing consultant?
The primary difference is that a fractional CMO is an executive who integrates into your leadership team to own marketing outcomes, while a consultant typically advises on strategy without owning execution. A fractional CMO manages the marketing budget, leads the team (internal or external), and is held accountable for growth targets like MRR. A consultant delivers a plan or audit and then disengages.
Limitations, Alternatives & Professional Guidance
While highly effective for many growing startups, the fractional model is not a fit for every organizational stage. For example, very large, enterprise-level HealthTech firms with multiple product lines typically require a full-time, in-house CMO for dedicated, day-to-day operational management and cross-departmental alignment. Additionally, the success of this approach depends heavily on the internal team’s ability to collaborate effectively with a remote executive leader and execute on high-level strategic directives.
Organizations should evaluate alternative approaches based on their specific funding stage and operational needs. A full-time CMO remains the traditional choice for well-funded, later-stage companies needing a dedicated internal leader. A specialized agency may be an option for companies that need execution in a single area (e.g., medical SEO or clinical content writing) but already have a strong internal marketing leader. Finally, an in-house team build-out is suited for founders who prefer to build and manage their own marketing team from the ground up, though this can be slower and more expensive.
It is crucial to recognize that implementing HIPAA-compliant technology and navigating advertising rules involves significant legal and technical risk. The FTC provides strict guidelines on digital advertising, emphasizing the need for clear and conspicuous disclosures, which is critical for HealthTech companies making claims about clinical efficacy.[5] We strongly recommend that any HealthTech company consult with both legal counsel specializing in healthcare law and a qualified MarTech architect before launching new marketing initiatives. This guide is for informational purposes and is not a substitute for professional legal or compliance advice.
Conclusion
In summary, engaging a fractional cmo for healthcare is often a strategic imperative for US startups needing to balance aggressive T2D3 growth with strict HIPAA compliance. Success in this complex sector relies on three key pillars: adapting growth frameworks for long sales cycles, architecting a compliant Signal Gateway to protect patient data, and positioning marketing operations as “valuation insurance” for future fundraising rounds. While the right approach depends heavily on a company’s specific stage and goals, integrating these methodologies can contribute to a highly resilient go-to-market strategy and serves as a core component of effective `healthcare marketing consulting`.
Algocentric Digital specializes in this exact integration of executive strategy and technical execution for HealthTech SaaS companies. If you are a HealthTech founder looking to implement a predictable growth engine while navigating regulatory complexities, you can explore our approach further. Download the HealthTech GTM Playbook to see these frameworks in action and discover how specialized marketing leadership can support your next phase of growth.
References
- SBA Office of Advocacy Report on AI Adoption. U.S. Small Business Administration (Research/Data). According to a 2025 report from the SBA’s Office of Advocacy, the AI adoption rate among US small businesses rose to 8.8%, with a significant focus on leveraging AI for marketing automation.
- U.S. Census Bureau Business and Economy Statistics. U.S. Census Bureau (Research/Data). Data from the U.S. Census Bureau indicates that approximately 78% of organizations were utilizing AI in 2024.
- HHS.gov Office for Civil Rights (OCR) Guidance. U.S. Department of Health and Human Services (Regulatory Guidance). In a December 2022 bulletin, the HHS Office for Civil Rights clarified that tracking technologies that collect individually identifiable health information are governed by HIPAA rules.
- NIST AI Risk Management Framework (AI RMF 1.0). National Institute of Standards and Technology (Framework/Standard). Released in January 2023, providing a voluntary structure for organizations to map, measure, and manage risks associated with AI systems.
- FTC Online Advertising and Marketing Guidelines. Federal Trade Commission (Regulatory Guidance). The FTC provides guidelines on digital advertising, emphasizing the need for clear and conspicuous disclosures regarding clinical efficacy.

Sergey Solonenko is the founder of Algocentric Digital Consultancy, an active digital strategist and a fractional CMO for many B2B SaaS brands embracing digital transformation. At Algocentric Digital Sergey’s focus is on empowering every B2B SaaS brand who is looking to scale their demand generation program. Sergey’s digital marketing experience over the last 10 years has allowed him to become a digital evangelist focused on improving B2B SaaS demand generation programs and consulting on best practices around account based marketing, sales and marketing team alignment, setting up better lead qualification systems and improving user experience through personalization by aligning martech with key marketing KPIs that ladder up to faster MRR for B2B SaaS brands.






